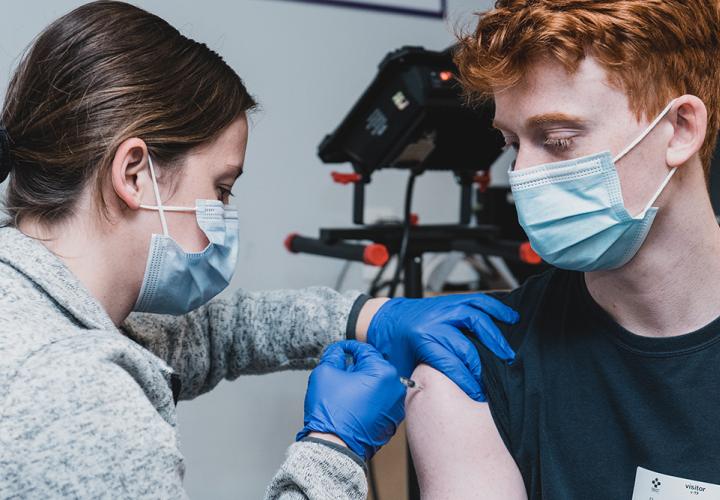
Yes – in fact, it appears to be even more effective, thanks to the strong immune response of younger adolescents.
> Related: Is Your Child Scared of Needles? Here’s How to Prepare for Their Vaccine
Is the Pfizer vaccine linked to any health risks in younger adolescents?
Rare cases of myocarditis and pericarditis have been reported in vaccinated teens and young people. Almost all of these cases have been mild, and gone away on their own. And a recent study shows that COVID-19 infection is actually much more likely to cause myocarditis than the vaccine.
There are no other known health risks from the vaccine. The FDA has carefully reviewed the data from the clinical trials in the 12 to 15 group and concluded that it’s safe for these ages, and the information from millions of individuals ages 16 and up who have already received the vaccine support this.
Will short-term side effects from the first or second shot (like a sore arm) be the same for 12- to 15-year-olds as older individuals?
After receiving any vaccine, it’s common to have short-term effects like a sore arm, fever, chills or headache in the next 24 hours. Not everyone experiences these symptoms, but when they do, it’s a normal part of the body’s immune response.
All of these effects are possible for younger adolescents who receive the COVID-19 vaccine. In Pfizer’s COVID-19 vaccine trials, fevers were slightly more common among the 12- to 15-year-olds than in older age groups.
If my child already had COVID-19, should they still get the vaccine?
Yes. Even though they’ll have some level of natural immunity, the vaccine provides additional protection. It’s possible to get COVID-19 more than once, so the vaccine helps prevent another infection.
Should any children NOT get the vaccine – for example, kids with certain underlying health conditions?
Almost every eligible child should get the vaccine. One rare exception is kids who have had a severe allergic reaction to a similar vaccine, but they are very few and far between.
But as always, if you have questions about your child and the COVID-19 vaccine, please reach out to your child’s pediatrician. We’re here to guide you.
What about younger adolescents and others who are currently undergoing chemotherapy. Is it safe for them to get the COVID-19 vaccine?
Yes. It’s possible that the vaccine might not be as effective for an individual with a weakened immune system, which can occur with chemotherapy. But the vaccine is safe, and offers at least some protection. That’s especially important for this group, who are at greater risk for severe illness from COVID-19.
Your child’s care team will always know best, so please reach out to your child’s doctor for help with their specific vaccine plan.
Besides Pfizer, are any other COVID-19 vaccines currently available for ages 12 and up?
Not yet. Moderna, Johnson & Johnson, Novavax and others have announced trials for their own vaccines, so it’s possible that we’ll have a vaccine for ages 12 and up from more than one company soon.
Is Connecticut Children’s offering the COVID-19 vaccine?
Yes! Visit our vaccine information page for the latest updates and resources.
The vaccine is the best way to protect your child from COVID-19 and its variants. It’s also crucial for our community to reach widespread immunity and return to the traditions and activities we cherish. Please schedule your child’s vaccine as soon as they’re eligible.